|
When I was a teacher at John McGregor Secondary School in Chatham, Ontario, I had some articles published in CHEM 13 NEWS of the University of Waterloo . Here is one that appeared in two parts in the December 1980, and in January 1981 issues:
Integrated Science
A unit for use by High School Science Students
Thomas A. Vella-ZarbJohn McGregor Secondary School, 300 Cecile Avenue, Chatham ON N7M 2C6
| HOURS | URINE VOLUME (cm3) | SPECIFIC GRAVITY |
| 1 | 140 | 1.008 |
| 2 | 500 | 1.002 |
| 3 | 270 | 1.003 |
| 4 | 90 | 1.010 |
| Total volume in the first four hours is 1000 cm3 6 | 40 | 1.020 |
| 8 | 45 | 1.026 |
| 10 | 85 | 1.025 |
| 12 | 30 | 1.031 |
| Time | Volume (cm3) | Specific gravity |
| 10.00 am | ||
| 12.00 noon | ||
| 2.00 pm | ||
| 4.00 pm | ||
| 6.00 pm | ||
| 8.00 pm | ||
| 8.00 pm to 8.00 am |
Usual values for the night sample are between 250 and 500 cm3. This value should never exceed 750 cm3 in a normal person. Its specific gravity should be about 1.018. During the day, one of the specimens should have a specific gravity over 1.018. The difference between the highest and lowest specific gravity findings should not be less than 8 to 9 points. (Note: Points in this case refer to the last two digits after the decimal.) If there is impairment of renal function, the night sample volume 750 cm3 or more would indicate nocturnal polyuria. Another indication of impaired renal
function is evident when the difference in the specific' gravity of the various samples is less than 8 or 9 points. In this case, the kidneys are losing their power to concentrate the urine. (The figures have to be adjusted if there is significant albumin or sugar present in the urine.) The subject takes a normal supper in the evening before the test, making sure that it contains a large amount of protein but not more than 200 cm3 of fluid. No more food or drink is then allowed until the end of the test the following morning. Before retiring, the subject empties his/her bladder and this specimen is discarded. The urine is then collected in three specimens as follows: A sample of urine is allowed to stand in a cylinder
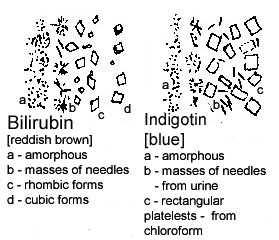
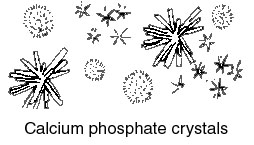
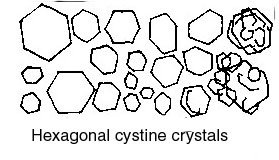
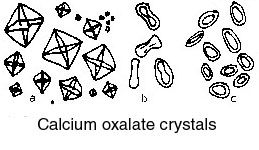
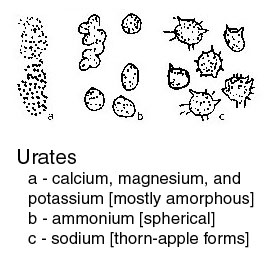
or a suitable sedimentation glass, and the sediment is then pipetted onto a slide for examination. If the urine appears to be quite clear, a sediment may be obtained more readily by centrifuging the sample. What one observes in the sediment may be grouped as follows.
DOCTORING THE SAMPLES OF URINEWe may assume that the samples of urine provided by the students will be normal since it is presumed that the students are healthy. To make the tests more interesting and varied, it may be advisable to "doctor" the specimens in different ways tb get positive reactions. A drop or two of acetone can easily be detected in the acetone test. Proteins are plentiful. Squeezing a little juice from some raw meat into the specimen or adding a tiny bit of egg white will give a guud positive result for the protein-in-urine test. When doctoring for sugars you must remember that the sugar test is for reducing sugars, and so dextrose, fructose, and lactose are to be added - not sucrose. PROTEINProtein or albumin is not normally present in urine. When albumin is found, the condition is known as albuminuria, and this indicates a flaw in the filtering system of the kidneys. Various tests are . available to detect albumin in the urine. Pour a sample of urine into a pyrex test tube
(about 10 cm3 is sufficient). Heat the upper portion of the sample over a flame. Pour about 5 cm3 of urine into a test tube. Add, drop by drop, a 20 to 25% solution of sulfosalicylic acid, and mix. If albumin is present, a white cloud will form. This turbidity is not cleared upon heating. This test is very sensitive, being able to detect protein in concentrations as low as 15 parts per million.
To 5 cm3 of filtered clear urine, add 0.5 cm3 Sorensen's acetate buffer solution and boil for about one-half a minute. A fine flocculation indicates the presence of more than traces of albumin. (Note: phosphates and urates do not give any flocculation with this test.) When Albumatest tablets are dissolved according to directions on the commercial package, they form a solution of sulfosalicylic acid, and sodium bicarbonate. Equal portions, usually about 10 drops, of urine and tho prepared solution are mixed together and a positive reading is made according to the amount of cloudiness or prrcipitation that forms. This test, when positive, is a significant diagnostic aid in multiple myeloma - a malignant tumour in the bone marrow.
Pour 10 cm3 urine into a test tube. Place this test tube and a thermometer into a beaker of water. Heat gently, observing both the urine and the thermometer. A positive Bence Jones is indicated by the following three conditions, all of which have to be met: SUGARSBENEDICT'S REAGENT To 5 cm3 of urine, add 0.5 cm3 of Benedict's reagent. Boil vigorously for about two minutes. Positive readings vary from yellow to brick red precipitation. ACETONE
The presence of acetone is very significant, because it often indicates acidosis. When acidosis is present in the body, the alkali reserve is decreased, and this may be fatal. Untreated diabetes mellitus, some fever, diarrhoea, excessive vomiting, and other conditions where the metabolism of glucose is impaired may be detected by the presence of acetone in the urine
Pour about 5 cm3 urine into a test tube. Add, drop by drop, loo w/v solution of ferric chloride until no more precipitate forms. In the presence of excessive quantities of diacetic acid (i.e., acetoacetic acid), a Bordeaux red colour will develop. If acetylsalicylic acid is present, a purple colour will develop, but the colour may be deceptive, and confusion may arise. To confirm the presence of diacetic acid, boil 5 cm3 of urine in a pyrex test tube for 3 minutes. (Alternatively, place it in boiling water for 15 minutes). Any diacetic acid present will
thus be converted to acetone, which will volatilize. After the sample cools, repeat the test with ferric chloride. If diacetic acid was originally present, the test should now be negative. If acetylsalicylic acid or similar drugs were present, the test will still be positive. BLOOD AND BLOOD PIGMENTSShould there be more than a few erythrocytes in the urine sediment, there is cause for concern, since their presence indicates bleeding within the urinary tract. The condition known as haemoglobinuria is present when occult (hidden) blood is present in the urine. This blood is not readily visible as it may be dissolved or haemolyzed. Excessive red cell destruction may occur because of a reaction during blood transfusion, as a result of severe burns, and in some chemical poisonings. These cause haemoglobinuria. In this test the haemoglobin in the blood reacts with hydrogen peroxide liberating oxygen, which then reacts with an organic reagent producing a coloured compound. Mix the urine and pour about 2 cm3 into a test tube. Add 3 cm3 of a saturated solution of benzidine in glacial acetic acid and mix. Add 1 cm3 of a 3% solution of hydrogen peroxide and mix. A positive reaction is indicated by a green to blue colour. This is a convenient method, wherein the chemicals orthotoluidine and hydrogen peroxide are present in cellulose strips. The test is carried out by dipping the strip in a well mixed sample of urine, or briefly passing it through the urine stream. Reading is done after 30 seconds and the amount of occult blood present is estimated by comparison with the colour charts supplied by the manufacturers. This is similar to the previous test and is manufactured by the same company. The chemicals are contained in tablets. Place one tablet on a filter paper. Place
a large drop of urine on the tablet (so that the urine just flows over the sides.) A blue colour is a positive test. Again, the amounts may be read by comparison with the colour chart on the side of the container. DRY WEIGHT OF URINEStudents working with volumes and specific gravity
or urine samples should be encouraged to find out the dry weight of urine. A normal adult excretes 55 - 70 g of solids in a twenty-four hour period. Obviously, one method of determining the dry weight would be experimentally by actually evaporating to dryness samples of urine, and then calculating from the results they find. They may be interested in a simple formula devised by Hoppe-Seyler: ANALYSING URINARY CALCULISamples of "stones" may be obtained from hospitals, [or veterinarians]
if you have the right connections. Students are usually able to furnish some samples from relatives who have had them removed, either by surgery, or by passing
them naturally (and painfully).
After all observations have been made on the intact stone, the stone is prepared for chemical examination. The whole stone or part thereof is finely pulverized in a mortar with a pestle. The first test to be performed is usually a flame test. This is carried out in either of two ways.
In this method, a nichrome or platinum innoculation loop is first dipped in distilled water and then to the finely powdered calculus. The powder covered loop is then placed in an open colourless flame and observed. In summary the observations and their interpretations are as follows: A small amount of the powder is transferred to a crucible or to a platinum boat. This container is held in the flame of a Bunsen burner and observations made as above.
Put some of the residual ash from the crucible method into a test tube and add either dilute hydrochloric acid, or add 2 cm3 distilled water and 2 drops of concentrated hydrochloric acid. Effervescence indicates the presence of oxalates. If no residue remains after igniting the powder, the oxalate test may be carried out later. Fill the test tube from above test with a saturated solution of ammonium oxalate. A heavy white precipitate indicates calcium. (Note: calcium is always present when oxalates are present, but the reverse is not always true,) To a small amount of unignited powder add a few drops of dilute HCl. Effervesence indicates the presence of carbonates. To the test tube from the above test add either of the following: To some unignited powder add 10 drops of 10% aqueous sodium hydroxide. The odour, if any, is noted. An odour of ammonia indicates the presence of ammonium magnesium phosphate.
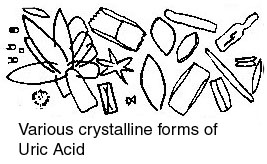
To some of the powdered stone in an evaporating dish add a drop of two of concentrated nitric acid. Strong foaming is presumed to indicate uric acid. To confirm this, evaporate to dryness - very carefully - over a steam bath. Cool to room temperature - the residue should be either yellow or red - and add two drops of concentrated ammonium hydroxide. If the colour turns to purplish-red, and fades when the crucible is heated again, this is definite proof of the presence of uric acid. Either of two tests may be performed: One drop of ''slurry'' from the carbonate test above is placed on a ceramic tile. Add one drop of a nitrophenylazobenzene alcoholic alkaline solution and five drops of sodium hydroxide solution. A blue precipitate indicates the presence of magnesium. To some powdered stone add about five drops of chloroform. A blue colour shows indigo. Dissolve some powdered stone in nitric acid. Evaporate to dryness un a water bath. Add three drops of ammonium hydroxide solution. If the residue turns orange, it
is a positive test for xanthine. (Compare this with the uric acid test above.)
Some unignited powder and a few drops of dilute hydrochloric acid are mixed together. Put in two drops of manganese oxide suspension in dilute hydrochloric acid, Tiny bubbles evolve within five minutes for a positive test. REAGENTSAlthough most of the reagents are readily available
in the laboratory, a number of them have to be freshly prepared according to special recipes.
Pipette 5 cm3 glacial acetic acid into a 100 cm3 volumetric flask, dilute to volume with distilled water and mix throughly. This is readily obtained from stock bottles. Dissolve 173 g sodium citrate, and 100 g anhydrous sodium carbonate in about 600 cm3 distilled water. In a separate container dissolve 17.3 g crystalline
cupric sulphate in about 100 cm3 distilled water. The copper sulphate solution is then added to the other solution with constant stirring. Filter if not clear. Make up to 1 litre with distilled water. Weigh 4 g benzidine powder and transfer to a brown bottle. Add 50 cm3 of glacial acetic acid. Shake to dissolve as much as possible. This solution is
stable for about one month. Put 10 g of anhydrous calcium chloride into a 100 cm3 volumetric flask. Dilute and make up to volume with distilled water. Pour 150 cm3 distilled water into a brown bottle. Add 10 g paradimethylaminobenzaldehyde. Swirl the mixture, and then carefully add 150 cm3 concentrated hydrochloric acid. Mix. Weight 200 g of sodium sulphate decahydrate and transfer to a litre volumetric flask into which you have previously poured 800 cm3 distilled water. Dissolve by swirling
the flask. Add 50 g sulfosalicylic acid and dilute to volume with distilled water. Mix. Weigh 10 g ferric chloride and transfer to a 100 cm3 volumetric flask. Make up to volume with distilled water, mixing thoroughly. Transfer 25 g trichloracetic acid to a flask containing 100 cm3 distilled water. Add 10 cm3 ferric chloride solution (see above). Mix. Weigh 5 g Iodine crystals and 10 g potassium iodide. Transfer to a brown bottle. Add 100 cm3 distilled water, and mix. Dissolve 4 g orthotoluidine in 100 cm3 glacial acetic acid. This solution keeps for about one month if kept in a brown bottle stored in a refrigerator. Add 40 g sodium nitroprusside (sodium nitroferricyanide) to 100 cm3 distilled water in a brown bottle. Shake
to dissolve as much as possible. Leave any undissolved salt in the bottle to ensure a saturated solution. Weigh 2.5 g ammonium oxalate and 2.5 g oxalic acid. Transfer to a brown bottle. Add 145 cm3 distilled water, and 5 cm3 glacial acetic acid. Mix well. Place ethyl alcohol in a beaker and add zinc acetate with constant stirring until no more dissolves. Dissolve 30 g sodium bisulphite in 200 cm3 distilled water. Dissolve 20 g sodium sulphite in 100 cm3
distilled water. Filter separately. Put 195 cm3 of the bisulphite solution in a flask. Add 0.5 g aminonaphtholsulphate and 5 cm3 of the sodium sulphite solution. Stopper and shake well. If the
salt does not dissolve, continue adding one cm3 of
the sulphite solution at a time and shake until it all dissolves. Store in a cool dark place. Stable for one month. To use, equal parts of I and II are freshly mixed together. | ||||||||||||||||||||||||||||||||||||||||||
Other Science Items of Interest
Anatomy Quiz
Chemistry Quiz
Interesting Chemistry Facts
A Fascinating Look at The Elements